Pages
Health Care News
Categories
- Asthma education
- Autism
- Canadian Health&Care Mall
- Cardiac function
- Critical Care Units
- Follicle
- Health
- health care medical transport
- health care programs
- Health&Care Professionals
- Hemoptysis
- Hormone
- Isoforms
- Nitroglycerin Patches
- Profile of interleukin-10
- Progesterone
- Pulmonary Function
- Sertoli Cells
- Theophylline
- Tracheoesophageal Fistula
Category Archives: Sertoli Cells
Disturbed Expression of Sox9 in Pre-Sertoli Cells: DISCUSSION(4)
Furthermore, in the present study Sox9 transcripts were detected by RT-PCR in both ovotestis and ovaries of B6.Ytir embryos at all developmental stages. This finding suggests that failure to up-regulate Sox9 in pre-Sertoli cells may be posttranscriptional of either Sry, Sox9, or both. Assuming that cells immunopositive to Sox9 detected at 11.5 and 12.5 dpc in B6.Ytir genital ridges are pre-Sertoli cells, current results indicate that 13.5 dpc represents a critical stage for the developmental fate of these gonads.
(more…)
Disturbed Expression of Sox9 in Pre-Sertoli Cells: DISCUSSION(3)
 A regional distribution of cells that express Sox9 in undifferentiated B6.Ytir gonads was observed. Sox9-positive cells appeared in the middle region, whereas they were absent from the cranial and caudal regions. Furthermore, cross sections of genital ridges at 11.5 and 12.5 dpc showed Sox9-positive cells among the innermost cells and rarely among coelomic epithelial cells. This distribution was similar in all embryos at 11.5 and 12.5 dpc, suggesting that regional location of pre-Sertoli cells in early genital ridges does not preclude their fate to differentiate as ovotestes or ovaries.
(more…)
A regional distribution of cells that express Sox9 in undifferentiated B6.Ytir gonads was observed. Sox9-positive cells appeared in the middle region, whereas they were absent from the cranial and caudal regions. Furthermore, cross sections of genital ridges at 11.5 and 12.5 dpc showed Sox9-positive cells among the innermost cells and rarely among coelomic epithelial cells. This distribution was similar in all embryos at 11.5 and 12.5 dpc, suggesting that regional location of pre-Sertoli cells in early genital ridges does not preclude their fate to differentiate as ovotestes or ovaries.
(more…) Disturbed Expression of Sox9 in Pre-Sertoli Cells: DISCUSSION(2)
In B6.Ytir gonads that take the ovarian pathway, Sertoli cells expressing Sox9 protein disappeared beyond 13.5 dpc; we did detect Sox9 transcripts in B6.Ytir ovaries, but at significantly lower levels than in gonads that form seminiferous cords (at 17.5 dpc). Since Sox9 transcripts in B6.Ytir ovaries are lower than in B6 or CD1 testes, our results indicate that maintenance of Sox9 transcripts at threshold levels may be necessary for pre-Sertoli cells to undergo further differentiation.
(more…)
Disturbed Expression of Sox9 in Pre-Sertoli Cells: RESULTS(5)
At 30 dpp as at 18.5 dpc, the levels of Sox9 appeared similar in testes of CD1 and B6 strains. In contrast to fetal testes, B6.Ytir gonads that developed as testes or bilateral ovaries did not show significant differences in Sox9 transcript levels compared with those in B6 testes. However, B6.Ytir ovaries of hermaphrodite B6.Ytir mice showed significantly lower levels than B6.Ytir females with bilateral ovaries (Fig. 6B). Finally, at 60 dpp the levels of Sox9 were not significantly different between B6 and B6.Ytir testes. In contrast, in B6.Ytir ovaries, Sox9 transcripts were no longer detected (Fig. 6C).
(more…)
Disturbed Expression of Sox9 in Pre-Sertoli Cells: RESULTS(4)
 Table 1 summarizes our observations of Sox9-positive cells in B6.Ytir embryos at various developmental stages. Although in 50% both gonads and in 25% one gonad showed pre-Sertoli cells at 13.5 dpc, in 25% both gonads and in 25% one gonad showed Sox9-positive Sertoli cells by 18.5 dpc. Taken together, Sox9-positive cells were absent in about half of the gonads studied between 13.5 and 18.5 dpc. This corresponds to the reported proportion of ovotestis and ovaries that form in B6.Ytir mice.
(more…)
Table 1 summarizes our observations of Sox9-positive cells in B6.Ytir embryos at various developmental stages. Although in 50% both gonads and in 25% one gonad showed pre-Sertoli cells at 13.5 dpc, in 25% both gonads and in 25% one gonad showed Sox9-positive Sertoli cells by 18.5 dpc. Taken together, Sox9-positive cells were absent in about half of the gonads studied between 13.5 and 18.5 dpc. This corresponds to the reported proportion of ovotestis and ovaries that form in B6.Ytir mice.
(more…) Disturbed Expression of Sox9 in Pre-Sertoli Cells: RESULTS(3)
Whereas in the well-organized seminiferous cords the germ cells entered the mitotic resting stage (Fig. 3A), in the ovarian regions they entered the prophase of the first meiotic division as evidenced by the synaptonemal complexes clearly detected with the electron microscope (Fig. 3B).
(more…)
Disturbed Expression of Sox9 in Pre-Sertoli Cells: RESULTS(2)
 Sox9-positive cells were located in the medial region of the gonad where they organize the incipient seminiferous cords together with the germ cells (Fig. 1C). In the B6.Ytir genital ridges showing Sox9-positive cells at 13.5 dpc, their number and the area of distribution along the medial region showed individual variations. Some gonads had scant randomly distributed Sox9-positive cells, whereas others revealed only a few at the mesonephric-gonad boundary (Fig. 1, D and E).
(more…)
Sox9-positive cells were located in the medial region of the gonad where they organize the incipient seminiferous cords together with the germ cells (Fig. 1C). In the B6.Ytir genital ridges showing Sox9-positive cells at 13.5 dpc, their number and the area of distribution along the medial region showed individual variations. Some gonads had scant randomly distributed Sox9-positive cells, whereas others revealed only a few at the mesonephric-gonad boundary (Fig. 1, D and E).
(more…) Disturbed Expression of Sox9 in Pre-Sertoli Cells: RESULTS(1)
Timing and Distribution of Pre-Sertoli Cells in B6. Ytir Fetal Gonads
Pre-Sertoli cells were identified by immunofluorescent staining with an antibody against Sox9 protein and were observed in all 12 B6.Ytir gonads at 11.5 and 12.5 dpc (Table 1). Longitudinal and cross-serial sections of urogenital ridges revealed a distribution pattern of pre-Sertoli cells within the still undifferentiated genital ridges. Cross serial sections revealed that pre-Sertoli cells were located among the innermost cells of the thickening genital ridges and rarely formed part of the coelomic epithelium (Fig. 1A). Longitudinal sections showed that pre-Sertoli cells were located in the medial region, whereas they were absent from both the cranial and caudal regions of the genital ridges (Fig. 1B).
Interestingly, whereas at 13.5 dpc Sox9-positive cells were present in both gonads of six out of 12 serially sectioned B6.Ytir embryos, they were considerably diminished in one gonad of three of these embryos. In three other embryos both gonads lacked Sox9-positive cells, and in the other three embryos Sox9-positive cells were absent in one of the two gonads (Table 1).
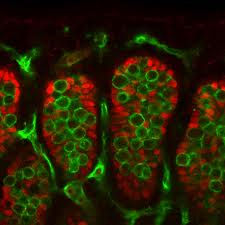
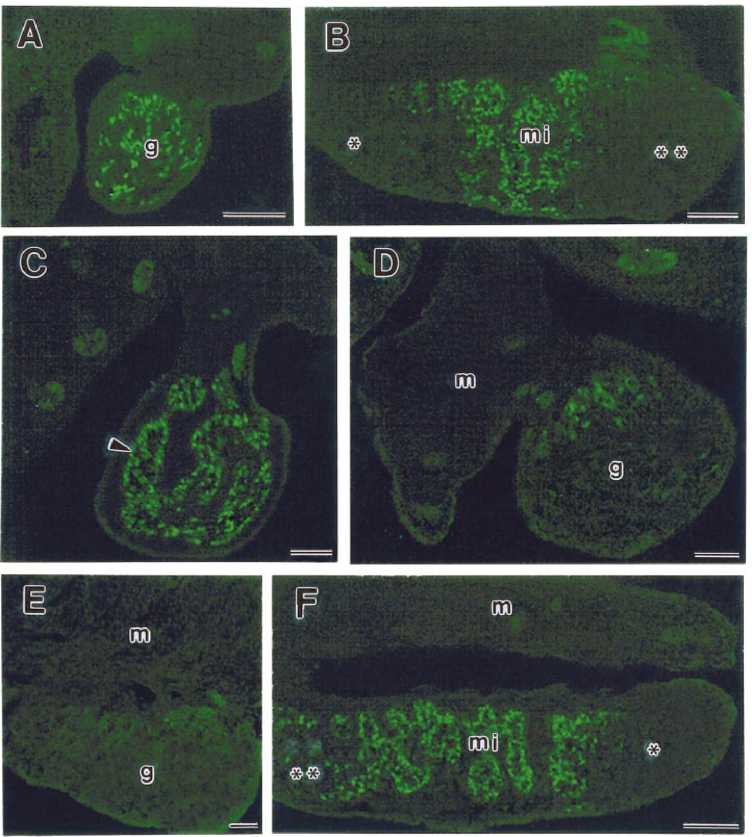 FIG. 1. Indirect immunofluorescent staining for Sox9 in frozen sections of B6.Ytir embryos at two developmental stages. A) and B) Urogenital ridges at 12.5 dpc. A) Cross-section of the middle region showing green-fluorescent Sox9-positive cells located in the core of the genital ridge (g). B) Longitudinal section showing Sox9-pos-itive cells located in the medial region (mi). Notice the absence in the cranial and caudal regions. C-F) gonads at 13.5 dpc. C) Cross section of the middle region showing that Sox9-positive cells are forming seminiferous cords (arrowheads). D) and E) Cross- and tangential sections of urogenital complexes showing few Sox9-positive cells in the gonad. These cells appear located in the dorsal side of the gonad (g) near the mesonephros (m). F) Longitudinal section showing organized seminiferous cords in the middle region (mi) of the gonad. Whereas in the cranial region several scattered Sox9-positive cells appear, in the caudal region they are absent. Bar = 100 ^m.
TABLE 1. SOX9 epression in serially sectioned gonads of B6.Ytir embryos.*
FIG. 1. Indirect immunofluorescent staining for Sox9 in frozen sections of B6.Ytir embryos at two developmental stages. A) and B) Urogenital ridges at 12.5 dpc. A) Cross-section of the middle region showing green-fluorescent Sox9-positive cells located in the core of the genital ridge (g). B) Longitudinal section showing Sox9-pos-itive cells located in the medial region (mi). Notice the absence in the cranial and caudal regions. C-F) gonads at 13.5 dpc. C) Cross section of the middle region showing that Sox9-positive cells are forming seminiferous cords (arrowheads). D) and E) Cross- and tangential sections of urogenital complexes showing few Sox9-positive cells in the gonad. These cells appear located in the dorsal side of the gonad (g) near the mesonephros (m). F) Longitudinal section showing organized seminiferous cords in the middle region (mi) of the gonad. Whereas in the cranial region several scattered Sox9-positive cells appear, in the caudal region they are absent. Bar = 100 ^m.
TABLE 1. SOX9 epression in serially sectioned gonads of B6.Ytir embryos.*
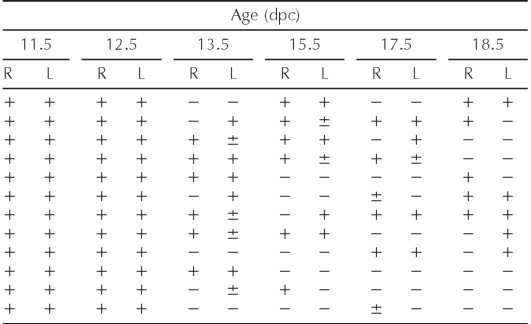 Right (R) and left (L) gonads. N = 12 B6. Ytir embryos at each age. +, ±, or — = abundant, few, or absent SOX9-expressing cells, respectively.
Right (R) and left (L) gonads. N = 12 B6. Ytir embryos at each age. +, ±, or — = abundant, few, or absent SOX9-expressing cells, respectively.
 FIG. 1. Indirect immunofluorescent staining for Sox9 in frozen sections of B6.Ytir embryos at two developmental stages. A) and B) Urogenital ridges at 12.5 dpc. A) Cross-section of the middle region showing green-fluorescent Sox9-positive cells located in the core of the genital ridge (g). B) Longitudinal section showing Sox9-pos-itive cells located in the medial region (mi). Notice the absence in the cranial and caudal regions. C-F) gonads at 13.5 dpc. C) Cross section of the middle region showing that Sox9-positive cells are forming seminiferous cords (arrowheads). D) and E) Cross- and tangential sections of urogenital complexes showing few Sox9-positive cells in the gonad. These cells appear located in the dorsal side of the gonad (g) near the mesonephros (m). F) Longitudinal section showing organized seminiferous cords in the middle region (mi) of the gonad. Whereas in the cranial region several scattered Sox9-positive cells appear, in the caudal region they are absent. Bar = 100 ^m.
TABLE 1. SOX9 epression in serially sectioned gonads of B6.Ytir embryos.*
FIG. 1. Indirect immunofluorescent staining for Sox9 in frozen sections of B6.Ytir embryos at two developmental stages. A) and B) Urogenital ridges at 12.5 dpc. A) Cross-section of the middle region showing green-fluorescent Sox9-positive cells located in the core of the genital ridge (g). B) Longitudinal section showing Sox9-pos-itive cells located in the medial region (mi). Notice the absence in the cranial and caudal regions. C-F) gonads at 13.5 dpc. C) Cross section of the middle region showing that Sox9-positive cells are forming seminiferous cords (arrowheads). D) and E) Cross- and tangential sections of urogenital complexes showing few Sox9-positive cells in the gonad. These cells appear located in the dorsal side of the gonad (g) near the mesonephros (m). F) Longitudinal section showing organized seminiferous cords in the middle region (mi) of the gonad. Whereas in the cranial region several scattered Sox9-positive cells appear, in the caudal region they are absent. Bar = 100 ^m.
TABLE 1. SOX9 epression in serially sectioned gonads of B6.Ytir embryos.*
 Right (R) and left (L) gonads. N = 12 B6. Ytir embryos at each age. +, ±, or — = abundant, few, or absent SOX9-expressing cells, respectively.
Right (R) and left (L) gonads. N = 12 B6. Ytir embryos at each age. +, ±, or — = abundant, few, or absent SOX9-expressing cells, respectively. Disturbed Expression of Sox9 in Pre-Sertoli Cells: MATERIALS AND METHODS(5)
 Ten microliters of each RT-PCR reaction were electrophoresed on a 2.5% agarose gel with 0.1 |xg/ml ethidium bromide in TAE 1X buffer. Gels were visualized by UV transillumination and photographed with a DS-34 Polaroid camera (Polaroid, Waltham, MA). The size of the amplified products was 310 bp for Sox9 and 243 for p-actin. The intensity of each band was quantified by densitometry using the Scion Image program. Results were expressed as arbitrary units of the ratio between Sox9 and p-actin mRNA levels.
In Situ Hybridization
Bar = 2.0 ^m.
In embryos at 12.5 and 13.5 dpp, the gonads were processed in situ attached to the dorsal body wall. At 18.5 dpc and 30 dpp, the gonads were dissected out and processed. A total of 11, 15, 7, and 11 gonads at 12.5 dpc, 13.5 dpc, 18.5 dpc, and 30 dpp, respectively, were processed. Whole-mount in situ hybridization for detection of Sox9 transcripts was carried out with probes kindly provided by Dr. Ramon Merino (Cantabria University, Spain). Reactions were performed in vitro using T3 or T7 RNA polymerase to generate sense and antisense RNA probes labeled with di-goxigenin-UTP. The complete procedure was as previously described by Wilkinson and Nieto. Reactions were developed with BCIP/NBT or with purple AP substrate (Boehringer-Mannheim, Roche, Indianapolis, IN).
Statistical Analysis
Profiles of Sox9 transcripts were quantified as mean ± standard deviation of at least three independent determinations. Variance between experiments was assessed by two-way ANOVA. The mean was used to evaluate the significant differences by paired Tukey test (P < 0.001).
Ten microliters of each RT-PCR reaction were electrophoresed on a 2.5% agarose gel with 0.1 |xg/ml ethidium bromide in TAE 1X buffer. Gels were visualized by UV transillumination and photographed with a DS-34 Polaroid camera (Polaroid, Waltham, MA). The size of the amplified products was 310 bp for Sox9 and 243 for p-actin. The intensity of each band was quantified by densitometry using the Scion Image program. Results were expressed as arbitrary units of the ratio between Sox9 and p-actin mRNA levels.
In Situ Hybridization
Bar = 2.0 ^m.
In embryos at 12.5 and 13.5 dpp, the gonads were processed in situ attached to the dorsal body wall. At 18.5 dpc and 30 dpp, the gonads were dissected out and processed. A total of 11, 15, 7, and 11 gonads at 12.5 dpc, 13.5 dpc, 18.5 dpc, and 30 dpp, respectively, were processed. Whole-mount in situ hybridization for detection of Sox9 transcripts was carried out with probes kindly provided by Dr. Ramon Merino (Cantabria University, Spain). Reactions were performed in vitro using T3 or T7 RNA polymerase to generate sense and antisense RNA probes labeled with di-goxigenin-UTP. The complete procedure was as previously described by Wilkinson and Nieto. Reactions were developed with BCIP/NBT or with purple AP substrate (Boehringer-Mannheim, Roche, Indianapolis, IN).
Statistical Analysis
Profiles of Sox9 transcripts were quantified as mean ± standard deviation of at least three independent determinations. Variance between experiments was assessed by two-way ANOVA. The mean was used to evaluate the significant differences by paired Tukey test (P < 0.001). Disturbed Expression of Sox9 in Pre-Sertoli Cells: MATERIALS AND METHODS(4)
Total RNA was resuspended in 12 |xl of DEPC-treated ddH2O. Reverse transcriptase polymerase chain reaction (RT-PCR) amplifications were performed using primers designed on the basis of the mouse Sox9 as previously reported. The sense primer was Sox9-1, 5'-GTG GCA AGT ATT GGT CAA-3'; the antisense primer was Sox9-2, 5'-GAA CAG ACT CAC ATC TCT-3'. For expression control, amplification of oligonucleotides used as internal controls were Actin 1 and Actin 2 as previously described.
(more…)
